
What is Arginase 1 Deficiency?
What is Arginase 1 Deficiency?
- ARG1-D is an ultra-rare, progressive, inherited metabolic disease caused by variants in the ARG1 gene that results in impaired or absent arginase 1 enzyme activity1,3
- Under normal conditions, arginine is hydrolyzed in the liver by the arginase 1 enzyme into ornithine and urea in the last step of the urea cycle1
- With impaired or absent enzyme activity, arginine and arginine-related metabolites, such as guanidino compounds, accumulate and are associated with neuromotor pathology1
- Symptomatic hyperammonemia and hyperammonemic crisis are less common in ARG1-D1
Elevated arginine is the hallmark feature of ARG1-D1
Persistently elevated levels of arginine in patients with ARG1-D are key drivers of disease manifestations, including progressive spasticity, developmental delay, and seizures.1
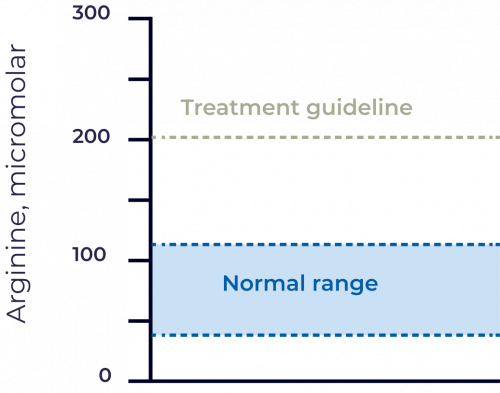
Current treatment guidelines for ARG1-D recommend lowering plasma arginine levels to reduce disease burden.1,5
Arginine Levels
The symptoms and progressive manifestations of ARG1-D are challenging and burdensome for patients and their families.6,7
The current standard of care for ARG1-D does not affect endogenous arginine production and is thus not sufficient for sustainably normalizing plasma arginine levels over time.1,8
The guideline-recommended target level for plasma arginine of <200 micromolar is above the upper limit of normal and may not be an optimal therapeutic target level for arginine.1,5
*The normal range for plasma arginine levels is 40-115 micromolar.1
Prevalence
ARG1-D may be more common than previously thought
Estimates of ARG1-D prevalence are limited by inconsistencies in newborn screening and high rates of misdiagnosis.10
- 1.5 estimated cases per million live births†
- ~250 patients (0.75 estimated cases per million people)
The clinical presentation of ARG1-D is heterogeneous3
Manifestations typically start in childhood, although symptoms can appear later in life due to accumulating levels of plasma arginine.3
Initial onset is typically confined to the lower limbs; however, as spasticity worsens, the upper limbs can also become affected.3
Persistently high plasma levels of arginine and arginine-related metabolites increase the risk of significant morbidity and early mortality.1
Symptoms often mimic other neurologic and neurometabolic disorders, which can result in misdiagnosis1,3
Progressive neurological symptoms
Family history
Onset and age of symptoms
Elevated plasma arginine
ARG1-D is a heterogeneous, progressively debilitating disease‡
References: 1. Diaz GA, et al. The role and control of arginine levels in arginase 1 deficiency. J Inherit Metab Dis. 2023;46(1):3-14. 2. Amayreh W, et al. Treatment of arginase deficiency revisited: guanidinoacetate as a therapeutic target and biomarker for therapeutic monitoring. Dev Med Child Neurol. 2014;56:1021-1024. 3. Carvalho DR, et al. Clinical features and neurologic progression of hyperargininemia. Pediatr Neurol. 2012;46:369-374. 4. Schlune A, et al. Hyperargininemia due to arginase I deficiency: the original patients and their natural history, and a review of the literature. Amino Acids. 2015;47:1751-1762. 5. Häberle J, et al. Suggested guidelines for the diagnosis and management of urea cycle disorders: First revision. J Inherit Metab Dis. 2019;1-39. 6. Olofsson S, et al. Disease burden among patients with Arginase 1 deficiency and their caregivers: A multinational, cross-sectional survey. J Inherit Metab Dis. 2024;65:450-460. 7. Olofsson S, et al. Societal costs and quality of life associated with arginase 1 deficiency in a European setting – a multinational, cross-sectional survey. J Med Econ. 2024;27(1):1146-1156. 8. Burrage LC, et al. Human recombinant arginase enzyme reduces plasma arginine in mouse models of arginase deficiency. Hum Mol Genet. 2015;24(22):6417-6427. 9. Bin Sawad A, et al. Epidemiology, methods of diagnosis, and clinical management of patients with arginase 1 deficiency (ARG1-D): a systematic review. Mol Genet Metab. 2022;137(1-2):153-163. 10. Therrell BL, et al. Newborn screening for hyperargininemia due to arginase 1 deficiency. Mol Gen Metab. 2017;121:308-313. 11. Catsburg C, et al. Arginase 1 deficiency: using genetic databases as a tool to establish global prevalence. Orphanet J Rare Dis. 2022;17(1):94. 12. Sun A, et al. Arginase deficiency. In: Adams MP, et al, eds. GeneReviews®. Seattle, WA: University of Washington, Seattle; 2020. 13. Sin YY, et al. Arginase-1 deficiency. J Mol Med (Berl). 2015;93:1287-1296. 14. McNutt MC, et al. Arginase 1 deficiency in patients initially diagnosed with hereditary spastic paraplegia. Mov Discord Clin Pract. 2022;10(1):109-114.
IMPORTANT SAFETY INFORMATION
See full prescribing information for complete boxed warning
Initiate LOARGYS in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue LOARGYS, and immediately initiate appropriate medical treatment, including use of epinephrine.
WARNINGS AND PRECAUTIONS
Hypersensitivity Reactions Including Anaphylaxis: Life-threatening hypersensitivity reactions, including anaphylaxis, have occurred in patients treated with enzyme replacement therapies, including LOARGYS. Hypersensitivity reactions that were mild to moderate in severity occurred in 13% (6/48) of LOARGYS-treated subjects in clinical trials. Hypersensitivity reactions have included facial swelling, rash, flushing and dyspnea. The reactions generally occurred with the first few doses but may occur later in treatment.
Administration of LOARGYS should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis in a healthcare setting with appropriate medical monitoring and support measures. Premedication with an antihistamine and/or corticosteroid should be considered in patients who previously have developed a hypersensitivity reaction. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue LOARGYS and immediately initiate appropriate medical treatment, including use of epinephrine. Consider the risks and benefits of re-administering LOARGYS in patients who have experienced a severe hypersensitivity reaction. Caution should be exercised upon rechallenge. Inform patients of the symptoms of life-threatening hypersensitivity reactions and to seek immediate medical attention should symptoms occur. If a mild or moderate reaction occurs, consider treatment with antihistamines and/or corticosteroids.
ADVERSE REACTIONS
The most common adverse reactions are vomiting, pyrexia, infusion associated reactions and constipation.
USE IN SPECIFIC POPULATIONS
Pregnancy: There are no available data on LOARGYS use in pregnant females to evaluate for a drug-associated risk of major birth defects, miscarriage or other adverse maternal or fetal outcomes.
Lactation: There is no data on the presence of LOARGYS in either human or animal milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for LOARGYS and any potential adverse effects on the breast-fed infant from LOARGYS or from the underlying maternal condition.
Pediatric: The safety and effectiveness of LOARGYS have been established for the reduction of plasma arginine in pediatric patients 2 years and older with ARG-1 D, in conjunction with dietary protein restriction. The safety and effectiveness of LOARGYS have not been established for the reduction of plasma arginine in pediatric patients aged less than 2 years with ARG-1 D.
Geriatric: Clinical studies of LOARGYS did not include subjects 65 years of age and older to determine whether they respond differently from younger adult subjects.
Indication
LOARGYS is an arginine specific enzyme indicated for the treatment of hyperargininemia in adult and pediatric patients 2 years of age and older with Arginase 1 Deficiency (ARG1-D), in conjunction with dietary protein restriction.
This indication is approved under accelerated approval based on reduction of plasma arginine. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.