
Dosing and Administration
Dosing & Administration
- Administer LOARGYS under the supervision of a healthcare provider knowledgeable in the management of hypersensitivity reactions, including anaphylaxis
- Initiate LOARGYS in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment
- Consider pre-medication with antihistamines
- Obtain a baseline plasma arginine concentration
- LOARGYS is supplied in 2 mg/0.4 mL single-dose vials
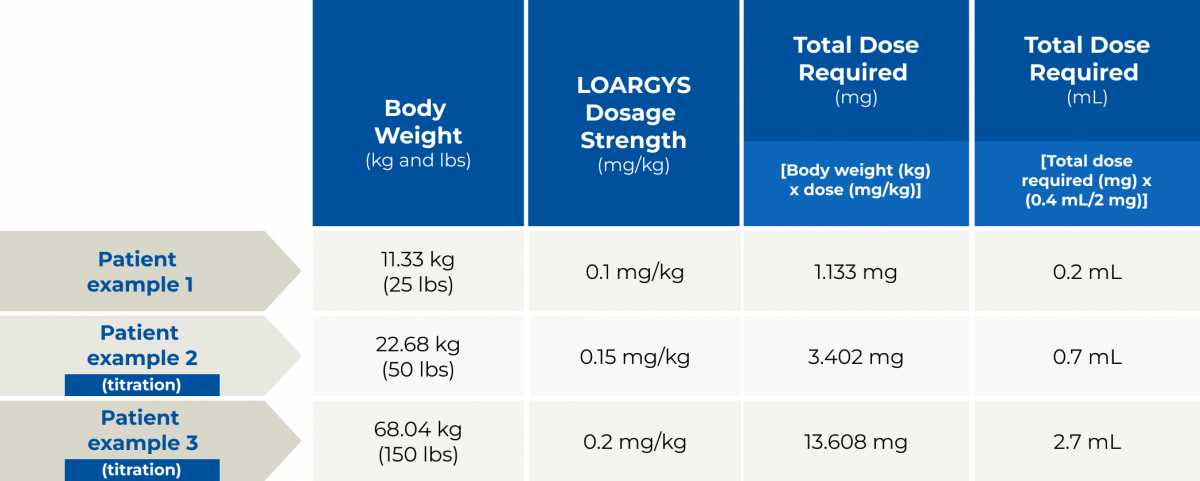
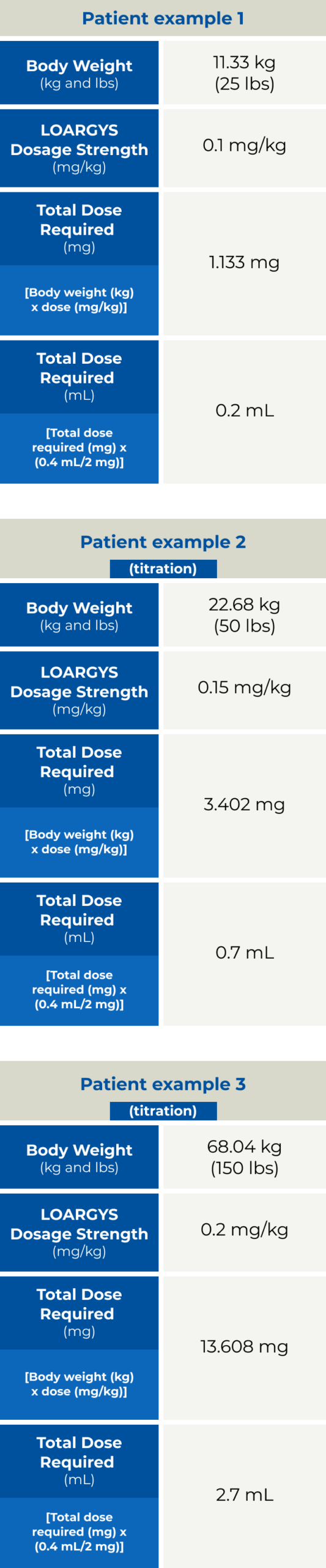
- The recommended starting dosage of LOARGYS is 0.1 mg/kg (actual body weight) administered via intravenous infusion once weekly
- Store refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Do not freeze. Do not shake
Please see full Prescribing Information for more directions on proper dosing and administration.
- For patients treated with LOARGYS, plasma arginine samples must be collected into Immedica Pharma’s Nor-NOHA Blood Collection Tubes, which contain Nω-hydroxy-nor-Arginine (nor-NOHA), an enzyme inhibitor used to inhibit post-sampling degradation of arginine by LOARGYS
- Plasma arginine concentration is then measured using Immedica Pharma’s LOARGYS Arginine Assay
After four weeks of LOARGYS administration, measure pre-dose (168 hours after prior dose) plasma arginine to determine the need for dosage adjustment. If two consecutive weekly pre-dose measurements of plasma arginine are not in the desired therapeutic range, increase or decrease the weekly LOARGYS dosage as follows:
- If the pre-dose plasma arginine concentration is below 50 micromolar, reduce the weekly LOARGYS dosage by 0.05 mg/kg
- If the pre-dose plasma arginine concentration is above 150 micromolar, increase the weekly LOARGYS dosage by 0.05 mg/kg
The maximum recommended LOARGYS dosage is 0.2 mg/kg once weekly. If a dose is missed, administer LOARGYS as soon as possible. Do not administer two LOARGYS doses on the same day or within four days of another dose to make up for a missed dose. Ensure there is a minimum of 4 days between doses.
Determine the number of LOARGYS vials needed based on actual body weight in kg and the recommended dose. Round the calculated volume of LOARGYS to nearest 0.1 mL.
LOARGYS preparation instructions
- Remove the vial(s) from the refrigerator and allow the vial(s) to reach room temperature
- Visually inspect the solution in the vials for particulate matter and discoloration. The solution should be clear to slightly opalescent, colorless to slightly yellow or slightly pink. Discard the vial(s) if the solution is not consistent with this appearance or if visible particulate matter is present
- Use a syringe to withdraw the calculated volume from the vial(s) and round the calculated volume of LOARGYS to nearest 0.1 mL
- Discard unused portion
- Dilute the withdrawn volume of LOARGYS solution in 0.9% Sodium Chloride Injection to a maximum concentration of 0.5 mg/mL
- Gently invert the infusion bag to mix the solution. Avoid vigorous shaking or agitation
- Administer the intravenous infusion over at least 30 minutes
- After LOARGYS infusion, use 0.9% Sodium Chloride to flush the line
- Do not mix other medications with LOARGYS or co-administer other drugs through the same intravenous line
- After 8 weeks of once weekly intravenous LOARGYS, patients may be switched to once weekly subcutaneous LOARGYS at the same dosage of intravenous therapy
- Administer the undiluted solution subcutaneously into the abdomen, lateral part of the thigh, or the side or back of the upper arms
- If more than 1 injection is needed for a single dose of LOARGYS, the injection sites should be at least 1 inch apart
- Do not inject into scar tissue or areas that are reddened, inflamed, or swollen. If injecting into the abdomen, avoid the area directly surrounding the navel
- Rotate injection sites between doses
- Patients treated subcutaneously may experience shorter periods of hypoargininemia than patients treated with LOARGYS intravenously
- If patients tolerate maintenance subcutaneous administration of LOARGYS, they may receive subcutaneous administration at home under the supervision of a healthcare provider
When switching patients from subcutaneous administration in a supervised clinical setting to at home administration under the supervision of a healthcare provider, initially use the same dose
- Use LOARGYS immediately after preparation. If not used immediately, store the diluted solution for intravenous use in the infusion container and the undiluted solution for subcutaneous use in the syringe for up to:
- 2 hours at room temperature at 20°C to 25°C (68°F to 77°F) or
- 4 hours if stored refrigerated at 2°C to 8°C (36°F to 46°F)
- Discard LOARGYS if not administered within these time frames, including total infusion time, if administered intravenously
Reference: LOARGYS (pegzilarginase-nbln) Prescribing Information. Immedica Pharma.
IMPORTANT SAFETY INFORMATION
See full prescribing information for complete boxed warning
Initiate LOARGYS in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue LOARGYS, and immediately initiate appropriate medical treatment, including use of epinephrine.
WARNINGS AND PRECAUTIONS
Hypersensitivity Reactions Including Anaphylaxis: Life-threatening hypersensitivity reactions, including anaphylaxis, have occurred in patients treated with enzyme replacement therapies, including LOARGYS. Hypersensitivity reactions that were mild to moderate in severity occurred in 13% (6/48) of LOARGYS-treated subjects in clinical trials. Hypersensitivity reactions have included facial swelling, rash, flushing and dyspnea. The reactions generally occurred with the first few doses but may occur later in treatment.
Administration of LOARGYS should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis in a healthcare setting with appropriate medical monitoring and support measures. Premedication with an antihistamine and/or corticosteroid should be considered in patients who previously have developed a hypersensitivity reaction. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue LOARGYS and immediately initiate appropriate medical treatment, including use of epinephrine. Consider the risks and benefits of re-administering LOARGYS in patients who have experienced a severe hypersensitivity reaction. Caution should be exercised upon rechallenge. Inform patients of the symptoms of life-threatening hypersensitivity reactions and to seek immediate medical attention should symptoms occur. If a mild or moderate reaction occurs, consider treatment with antihistamines and/or corticosteroids.
ADVERSE REACTIONS
The most common adverse reactions are vomiting, pyrexia, infusion associated reactions and constipation.
USE IN SPECIFIC POPULATIONS
Pregnancy: There are no available data on LOARGYS use in pregnant females to evaluate for a drug-associated risk of major birth defects, miscarriage or other adverse maternal or fetal outcomes.
Lactation: There is no data on the presence of LOARGYS in either human or animal milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for LOARGYS and any potential adverse effects on the breast-fed infant from LOARGYS or from the underlying maternal condition.
Pediatric: The safety and effectiveness of LOARGYS have been established for the reduction of plasma arginine in pediatric patients 2 years and older with ARG-1 D, in conjunction with dietary protein restriction. The safety and effectiveness of LOARGYS have not been established for the reduction of plasma arginine in pediatric patients aged less than 2 years with ARG-1 D.
Geriatric: Clinical studies of LOARGYS did not include subjects 65 years of age and older to determine whether they respond differently from younger adult subjects.
Indication
LOARGYS is an arginine specific enzyme indicated for the treatment of hyperargininemia in adult and pediatric patients 2 years of age and older with Arginase 1 Deficiency (ARG1-D), in conjunction with dietary protein restriction.
This indication is approved under accelerated approval based on reduction of plasma arginine. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.