
Efficacy & Safety
Efficacy & Safety
LOARGYS clinical development program: demonstrated efficacy and safety in ARG1-D
A total of 48 patients aged 2-32 years (40 children and 8 adults) with a confirmed diagnosis of ARG1-D have received LOARGYS in clinical studies. Patients included in the LOARGYS clinical studies have received treatment for up to approximately 5 years.1-3
The LOARGYS clinical development program included the first-ever randomized, double-blind, and placebo-controlled phase 3 clinical study in ARG1-D, the Pegzilarginase Effect on Arginase 1 Clinical Endpoints (PEACE) study, to demonstrate the efficacy of pegzilarginase relative to placebo based on a statistically significant decrease in plasma arginine concentrations.1,4
PEACE: Phase 3 Study
Pegzilarginase Effect on Arginase 1 Clinical Endpoints (PEACE) was a pivotal phase 3, randomized, double-blind, placebo-controlled, international study that evaluated the efficacy and safety of weekly LOARGYS added to standard of care management1
Efficacy outcomes
Primary: Change from baseline in plasma arginine at week 24
| IV, intravenous; LTE, long-term extension; SC, subcutaneous; QW, once weekly. | |
| The associated clinical response of this product is not yet confirmed. | |
| * | ARG1-D diagnosis was based on elevated plasma arginine, pathogenic ARG1 variant, and/or diminished erythrocyte arginase activity. |
| † | Weekly dosing; if needed, dose modifications permitted based on plasma arginine levels with maintenance of blinding. |
| ‡ | Blinding maintained through the first 8 weeks of the LTE. |
PEACE: Patient Population
The patient population in PEACE was consistent with the early-phase study cohort, and representative of the wider ARG1-D population1,2
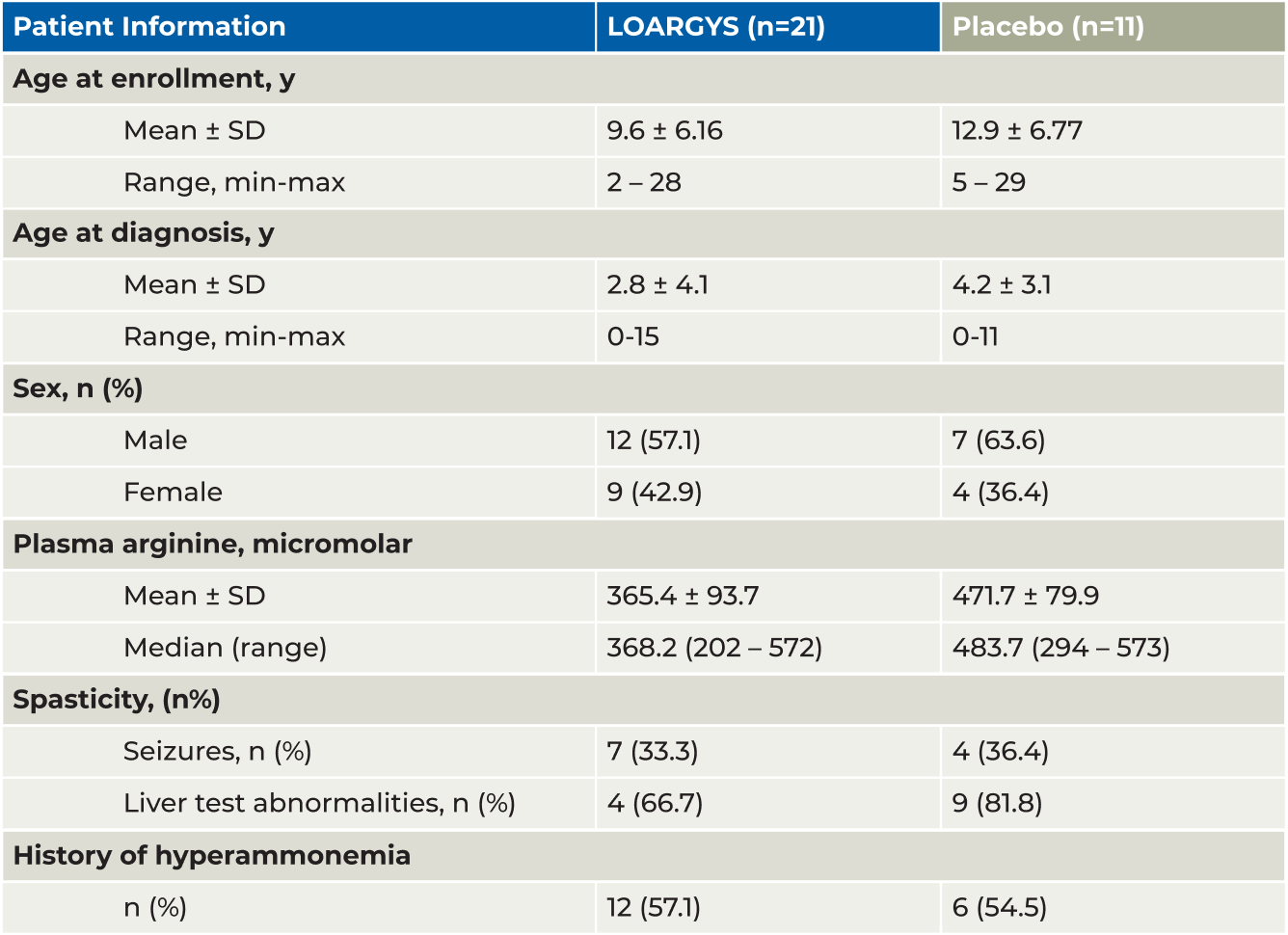
PHASE 3 EFFICACY RESULTS
- The median duration of LOARGYS exposure in the long-term extension (LTE), excluding the double-blind period of 24 weeks, was 94 weeks (range, 62-152 weeks)3
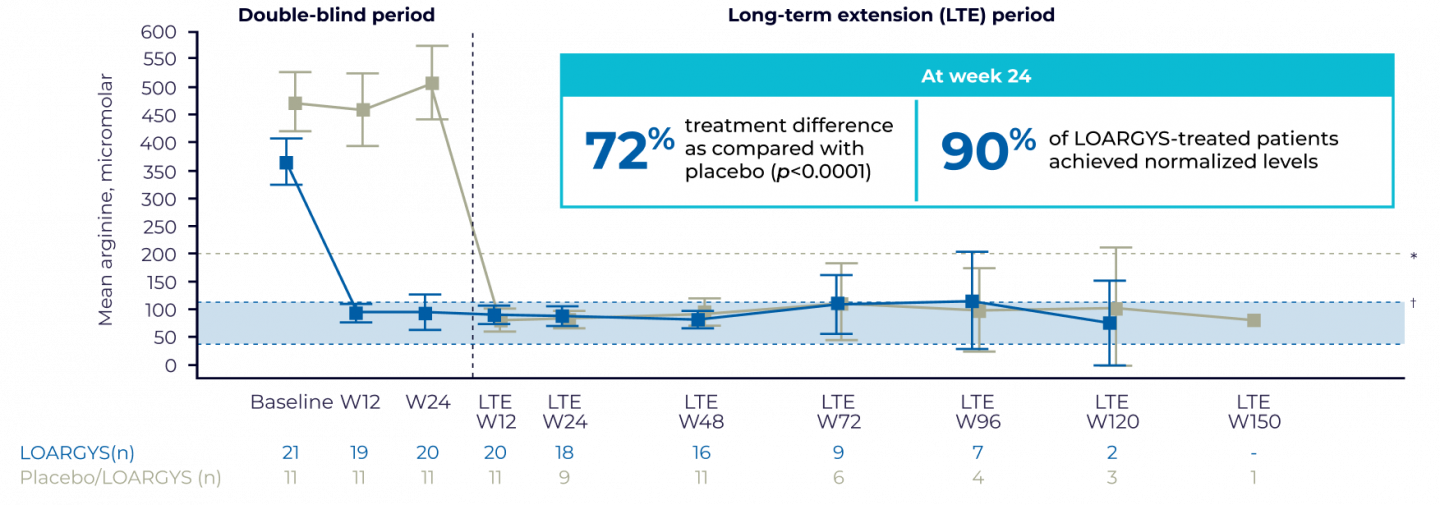
| * | The guideline-recommended target level for plasma arginine was <200 micromolar. |
| † | The normal range for plasma arginine (as defined in the PEACE study) was 40-115 micromolar. |
LOARGYS is well tolerated with demonstrated safety1,3§
- The safety of LOARGYS was evaluated in a phase 3 study (Trial 1, PEACE). Additional safety information was derived from Trial 2, a phase 1 open-label trial, and Trial 3, an open-label extension of Trial 21,3
- The most common adverse reactions are vomiting, pyrexia, infusion associated reactions, and constipation3
Hypersensitivity reactions (HSRs)
Injection site reactions (ISRs)
Immunogenicity
- There were no discontinuations due to adverse events across all clinical trials1,2,5
- HSRs were mild to moderate in severity3
- Across all 3 trials, the incidence of HSRs was 42% (5/12) in LOARGYS-treated patients who developed ADAs and 3% (1/36) in those who were ADA negative3
| § | See full Prescribing Information for additional Safety Information. |
Publication
References: 1. Russo RS, et al. Efficacy and safety of pegzilarginase in arginase 1 deficiency (PEACE): a phase 3, randomized, double-blind, placebo-controlled, multi-centre trial. EClinicalMedicine. 2024;68:102405. 2. Diaz GA, et al. Clinical effect and safety profile of pegzilarginase in patients with arginase 1 deficiency. J Inherit Metab Dis. 2021;44(4):847-856. 3. LOARGYS (pegzilarginase-nbln) Prescribing Information. Immedica Pharma. 4. Data on File. Immedica Pharma. 5. McNutt M, et al. Long-term efficacy and tolerability of pegzilarginase in arginase 1 deficiency: results of two international multicentre open-label extension studies. J Inherit Metab Dis. 2025;48(4):e70066. doi:10.1002/jimd.70066.
IMPORTANT SAFETY INFORMATION
See full prescribing information for complete boxed warning
Initiate LOARGYS in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue LOARGYS, and immediately initiate appropriate medical treatment, including use of epinephrine.
WARNINGS AND PRECAUTIONS
Hypersensitivity Reactions Including Anaphylaxis: Life-threatening hypersensitivity reactions, including anaphylaxis, have occurred in patients treated with enzyme replacement therapies, including LOARGYS. Hypersensitivity reactions that were mild to moderate in severity occurred in 13% (6/48) of LOARGYS-treated subjects in clinical trials. Hypersensitivity reactions have included facial swelling, rash, flushing and dyspnea. The reactions generally occurred with the first few doses but may occur later in treatment.
Administration of LOARGYS should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis in a healthcare setting with appropriate medical monitoring and support measures. Premedication with an antihistamine and/or corticosteroid should be considered in patients who previously have developed a hypersensitivity reaction. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue LOARGYS and immediately initiate appropriate medical treatment, including use of epinephrine. Consider the risks and benefits of re-administering LOARGYS in patients who have experienced a severe hypersensitivity reaction. Caution should be exercised upon rechallenge. Inform patients of the symptoms of life-threatening hypersensitivity reactions and to seek immediate medical attention should symptoms occur. If a mild or moderate reaction occurs, consider treatment with antihistamines and/or corticosteroids.
ADVERSE REACTIONS
The most common adverse reactions are vomiting, pyrexia, infusion associated reactions and constipation.
USE IN SPECIFIC POPULATIONS
Pregnancy: There are no available data on LOARGYS use in pregnant females to evaluate for a drug-associated risk of major birth defects, miscarriage or other adverse maternal or fetal outcomes.
Lactation: There is no data on the presence of LOARGYS in either human or animal milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for LOARGYS and any potential adverse effects on the breast-fed infant from LOARGYS or from the underlying maternal condition.
Pediatric: The safety and effectiveness of LOARGYS have been established for the reduction of plasma arginine in pediatric patients 2 years and older with ARG-1 D, in conjunction with dietary protein restriction. The safety and effectiveness of LOARGYS have not been established for the reduction of plasma arginine in pediatric patients aged less than 2 years with ARG-1 D.
Geriatric: Clinical studies of LOARGYS did not include subjects 65 years of age and older to determine whether they respond differently from younger adult subjects.
Indication
LOARGYS is an arginine specific enzyme indicated for the treatment of hyperargininemia in adult and pediatric patients 2 years of age and older with Arginase 1 Deficiency (ARG1-D), in conjunction with dietary protein restriction.
This indication is approved under accelerated approval based on reduction of plasma arginine. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
